Image: selvanegra/Getty Images
This article is part of Harvard Medical School’s continuing coverage of medicine, biomedical research, medical education and policy related to the SARS-CoV-2 pandemic and the disease COVID-19.
Physician-scientists at Harvard Medical School and Beth Israel Deaconess Medical Center are now enrolling patients in a clinical trial to evaluate a potential treatment of patients with COVID-19. Part of a multisite investigation, the trial is evaluating the safety and efficacy of sarilumab, a biologic medication already approved for adults with moderately to severely active rheumatoid arthritis, for the treatment of COVID-19.
Robert Hallowell, HMS assistant professor of medicine and medical director of pulmonary ambulatory services at Beth Israel Deaconess, is leading the phase 2/3 trial.
“While the symptoms of COVID-19 are mild for many, roughly 15 percent are at risk of developing an aberrantly robust immune reaction, which in its most severe form can lead to respiratory failure and, potentially, death,” said Hallowell. “The ability to stop this reaction in its tracks would be a major step forward in the management of COVID-19, both for infected individuals and for hospitals on the front line of this pandemic.”
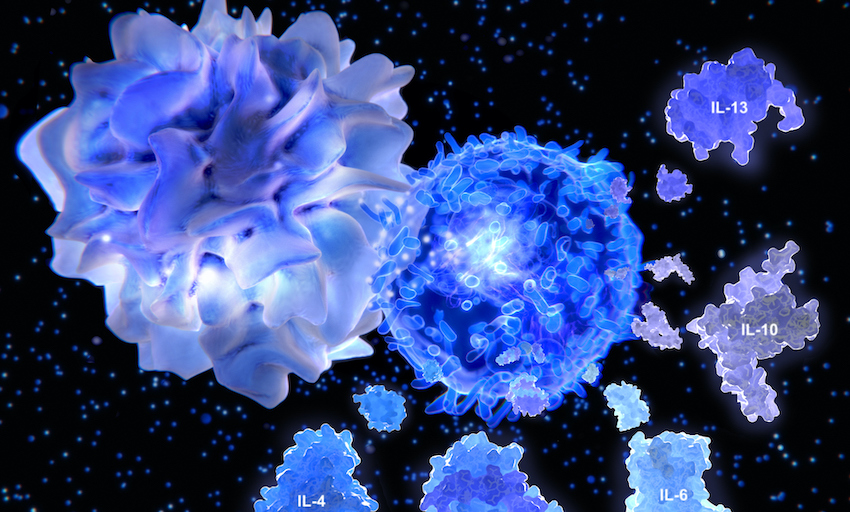
An inflammatory inhibitor, sarilumab was developed to block interleukin-6 (IL-6), an immune factor called a cytokine involved in the overactive inflammatory response that is a common feature of autoimmune disorders like rheumatoid arthritis.
The body releases cytokines, including IL-6, as part of its normal immune response to injured or infected tissues. In the average person, cytokines are responsible for symptoms such as redness and swelling around a wound or infected site. In patients with autoimmune disorders, cytokines can drive the immune response that attacks otherwise healthy tissues, such as cartilage in patients with rheumatoid arthritis or the cells of the pancreas in some people with type 1 diabetes.
Similarly, an overactive inflammatory response is thought to be the cause of the lung damage and respiratory distress in a significant percentage of patients with severe COVID-19. As the immune system releases cytokines to kill the virus, infected cells in the lungs become collateral damage. In turn, this injury to the lung tissues triggers additional inflammation, and the so-called cytokine storm begins to spiral out of control. The result can be lasting lung damage and scarring, organ failure or death.
Designed to halt this cytokine surge, sarilumab has the potential to improve outcomes for patients with severe cases of COVID-19, as well as reduce the expected demand for ventilators—at present the only course of treatment for acute respiratory failure—for patients with severe cases of COVID-19.
“We have enrolled eight patients since opening the trial and plan to enroll several patients a day until the trial ends as a way of offering this potential treatment to the community,” said Hallowell.
Hallowell’s clinical trial is one of several efforts fostered by the COVID-19 Innovation Hub, an effort led by Gyongyi Szabo, member of the HMS faculty of medicine and chief academic officer at Beth Israel Deaconess, to meet the emerging challenges of the COVID-19 pandemic. Other projects include research into a potential vaccine for the prevention of COVID-19, clinical trials to evaluate the antiviral medication remdesivir and the efficacy of an anticlotting medication for respiratory failure.
“As of now, patients with COVID-19 receive the gold-standard treatment of supportive care across our hospital system, but we continue to pursue new ways to care for them,” said Szabo. “If this drug can prevent patients from becoming gravely ill with COVID-19 or reduce the time it takes patients to recover, it could save lives.”
Adapted from a Beth Israel Deaconess news release.





