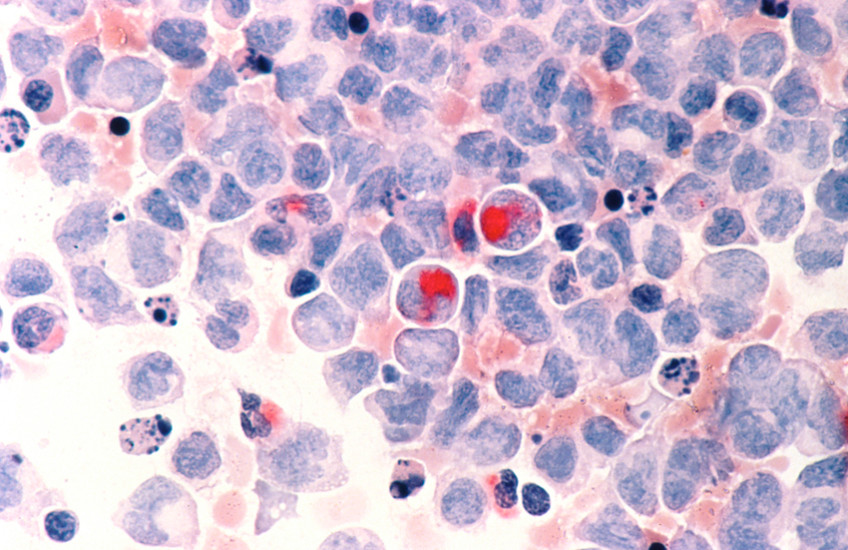
Every year, nearly 11,000 Americans die of acute myeloid leukemia, a blood cancer that affects mainly older adults. While most patients initially respond to chemotherapy, more than half of those who respond will eventually relapse as the cancer cells develop resistance to treatment.
In a new paper published today in Cell Research, a team of scientists led by Pier Paolo Pandolfi, the Victor J. Aresty Professor of Medicine at HMS and Beth Israel Deaconess Medical Center, reveal vulnerabilities in a subset of AML that could serve as the foothold researchers need to overcome resistance to therapy.
“There is an acute need to develop rational combinations to treat disease and overcome resistance that arises in response to therapy,” said Pandolfi, who is director of the Cancer Center and Cancer Research Institute at Beth Israel Deaconess. “By providing a critical understanding of the mechanisms underlying the disease, we identified a therapeutic strategy for treatment of this type of AML.”
About 20 percent of AML cases involve a mutation in a key metabolic enzyme called IDH (isocitrate dehydrogenase). Using a previously published genetically engineered mouse model and human cell-line in vitro and in vivo models for AML with this mutation, Pandolfi and colleagues identified key processes that these cancer cells use to develop—or evolve—drug resistance in response to therapies.
By following the pathway to resistance in a step-by-step manner, the scientists, including first author Vera Mugoni, a postdoctoral research fellow in the Pandolfi lab, identified critical vulnerabilities that researchers could potentially exploit for therapeutic benefit in all stages of the disease’s evolution. Moreover, the team demonstrated that a combination of drugs already in use for the treatment of another type of leukemia worked equally as well against this form of AML in both in vitro and in vivo settings.
“The molecular processes that are frequently required to promote resistance to therapy leave tumors vulnerable in other areas,” said Mugoni. “In the case of AML with mutation to IDH enzymes, both metabolism and differentiation pathways were altered, exposing vulnerabilities that become exacerbated as the leukemia evolves, and for which we already have approved agents that can be utilized.”
Another form of leukemia known as acute promyelocytic leukemia was once a fatal diagnosis. Now considered a curable disease, researchers, including Pandolfi, demonstrated that arsenic trioxide was effective against APL in combination with another drug called all-trans retinoic acid. In the current study, Pandolfi and colleagues tested the combination in both the mouse model of AML with IDH mutation and in human AML cells. They determined that, while the drugs target different vulnerabilities in the different forms of leukemia, the combination proved “powerfully and exquisitely effective” against this subset of AML cells.
“With my lab’s previous discovery that all-trans retinoic acid and arsenic trioxide is curative in the treatment of acute promyelocytic leukemia, witnessing the translation of this same therapy to possible therapeutic approaches for other forms of AML is extremely rewarding,” said Pandolfi.
He added that his team is currently working to develop clinical trials to evaluate the all-trans retinoic acid and arsenic trioxide combination for treatment of AML with mutation in IDH enzymes at multiple centers throughout the United States and beyond.
This work was supported by EMBO Long Term Fellowships for postdoctoral research (ALTF 1011-2014); the European Commission (LTFCOFUND2013, GA-20130609409); the Leukemia and Lymphoma Society Career Development Program (grant 5437-16); the Marie Curie Action Fellowship Mobility Program for post graduate scientists in the Life Sciences (C.I. 130011240012025124540); the Società Italiana di Biofisica e Biologia Molecolare; the Ludwig Center at HMS; and National Institutes of Health/National Cancer Institute (grants R35CA197529 and R01CA142874).
Disclosures: Pandolfi is a member of the scientific advisory board of Agios Pharmaceuticals. Pandolfi, Mugoni and co-authors Clohessy and Chen have filed (Dec. 1, 2017) U.S. Patent Application No. 62/593,761, “Arsenic Trioxide and Retinoic Acid Compounds for Treatment of IDH-Associated Disorders.” This application is still pending. The remaining authors declare no competing interests.
Adapted from a Beth Israel Deaconess news release.
Image: National Cancer Institute