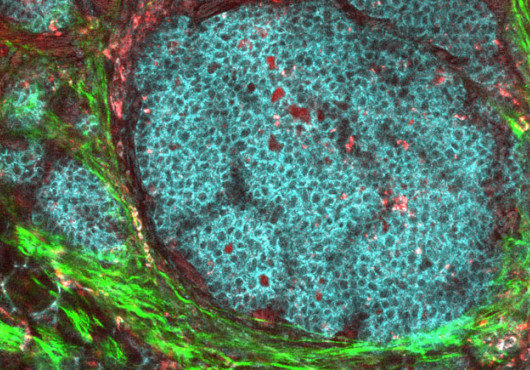
Single tenet has long undergirded stem cell and regenerative biology: for differentiated cells, their function is their fate, and only stem cells can be coaxed into a desired cell type. But new research from the lab of Harvard University Faculty of Arts and Sciences professor Douglas Melton rattles that foundational principle to the core. By converting mouse pancreatic exocrine cells into insulin-producing cells that are nearly identical to the beta cells destroyed by type 1 diabetes, Melton and his collaborators have demonstrated that even the most differentiated cells may be mutable.
If these findings can be applied to other cell types as well, then the future of regenerative biology may eventually bypass the burdensome and labor-intensive stem cell–based techniques.
“I think this could be applied to the nervous system or cardiovascular system,” said Melton at a press conference. “It represents a kind of parallel approach to how to make cells for regenerative medicine. This is a step forward toward eventually developing a treatment for diabetes.”
The findings were published online Aug. 27 in Nature.
Taking a cue from the work of Shinya Yamanaka of Kyoto University in Japan, who last year reprogrammed differentiated cells into pluripotent cells using a gene therapy technique, Melton and colleagues shuttled the genes for three transcription factors into the exocrine cells by attaching them to a virus that targets the pancreas. The three factors, Ngn3, Pdx1, and MafA, were identified by postdoctoral researcher Joe Zhou after two painstaking years of screening for genes that were essential to beta cell functioning.
The factors were injected directly into the pancreases of mice with diabetes. After only three days, approximately 20 percent of the affected exocrine cells began producing insulin, providing the mice with a profound therapeutic benefit.
Melton and his colleagues believe that these new cells are in every sense beta cells, not simply exocrine cells that produce insulin. The cells also make insulin granules for secretion, and they express a glucose transporter on their surface so they can measure blood glucose.
Ultimately, the researchers would like to see these cells migrate together to form pancreatic islets, something that did not occur in the animal models. Nevertheless, at the time of publication, the cells were still functioning, a timeframe that if translated into human years would equal roughly a decade. According to Melton, all evidence indicates that these cells simply “flipped” from exocrines to beta cells.
“We’ve looked for genes that are on in the stem cell and progenitor cell state and do not find them to be expressed,” said Melton, who is also co-director of the Harvard Stem Cell Institute and co-chair of the Department of Stem Cell and Regenerative Biology, which is run jointly by HMS and FAS. “But that’s negative evidence. The conversion happens very quickly, within three days. And in the production of things that involve reprogramming, it takes many rounds of cell division and weeks in culture. Here, it’s 20 percent of the cells quickly in three days.”
Melton suspects that this approach can apply to nervous system disorders and other conditions as well. For diseases such as amyotrophic lateral sclerosis, for example, he can envision switching astrocytes into healthy motor neurons, using genes that have yet to be identified.
As for direct clinical applications of his findings for diabetes in people, a likely approach might be to create the cells and the resulting islets in a dish, then transplant them into a patient’s pancreas or liver. (Direct injections into visceral organs can create complications.)
Joan Brugge, chair of the Department of Cell Biology at HMS, told the Harvard University News Office that the new study “provides exciting new insights into yet another aspect of cell plasticity that was not appreciated previously and that offers great potential therapeutically. Direct re-programming represents a more straightforward strategy to treat diseases involving loss of function of specific cell populations than approaches requiring an intermediate embryonic stem cell.”
Despite the excitement generated by these findings, Melton is adamant that research with human embryonic stem cells must continue. Embryonic stem cells, he says, provide a unique window into human health and disease. “ESC research will remain a key part of the long-term programs in regenerative medicine, and what we’re reporting [here] is another way of approaching the same kind of problem.”
Conflict Disclosure: The authors declare no conflicts of interest.
Funding Sources: Harvard Stem Cell Institute and the Howard Hughes Medical Institute