Can a single cell be sophisticated enough to "change its mind"? (Stentor footage courtesy: James Weiss (Jam's Germs), Bill Porter)
Once, single-cell life claimed sole dominion over the earth. For some three billion years, unfathomable generations of unicellular organisms ate, grew and reproduced among only each other. They evolved into predators and prey, thrived and spread across the primordial waters and land, and formed complex and dynamic ecosystems in every ecological niche on the planet. Around 600 million years ago, some even crossed the threshold into multicellularity.
Today, however, single-cell organisms are synonymous with notions like primitive and simple. Yet, new research suggests that they may be capable of much more than their very distant human cousins might suspect.
In an effort to replicate an experiment conducted over a century ago, systems biologists at Harvard Medical School now present compelling evidence confirming at least one single-cell organism—the strikingly trumpet-shaped Stentor roeselii—exhibits a hierarchy of avoidance behaviors.
Exposed repeatedly to the same stimulation—in this case a pulse of irritating particles—the organism can in effect “change its mind” about how to respond, the authors said, indicating a capacity for relatively complex decision-making processes.
The results are published online in Current Biology on Dec. 5.
“Our findings show that single cells can be much more sophisticated than we generally give them credit for,” said corresponding study author Jeremy Gunawardena, associate professor of systems biology in the Blavatnik Institute at HMS.
The researchers say such sophistication makes evolutionary sense.
“Organisms like S. roeselii were apex predators prior to multicellular life, and they are extremely widespread in many different aquatic environments,” he said. “They have to be ‘clever’ at figuring out what to avoid, where to eat and all the other things that organisms have to do to live. I think it’s clear that they can have complex ways of doing so.”
Fascinating yet forgotten
A decade ago, at a lecture by the English biologist Dennis Bray, Gunawardena was introduced to the work of the prominent American zoologist Herbert Spencer Jennings (Harvard A.B. 1895, Ph.D. 1896), who, in 1906, published the influential text Behavior of the Lower Organisms. One particular experiment caught Gunawardena’s eye.
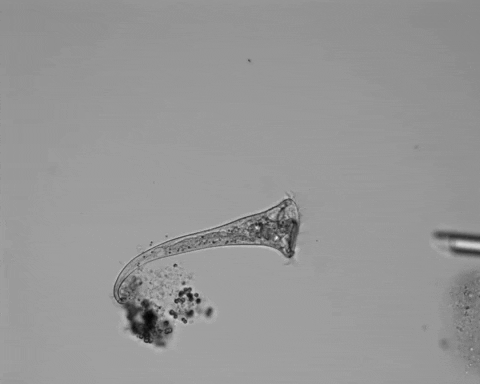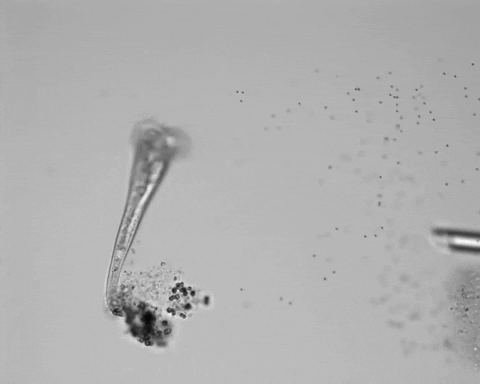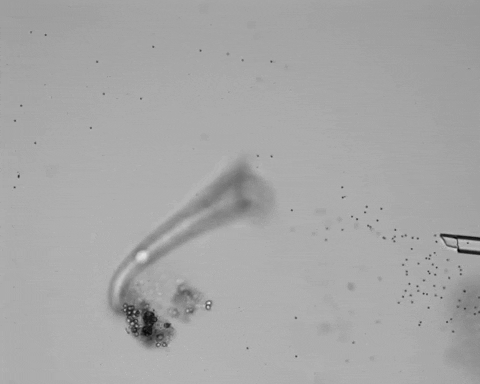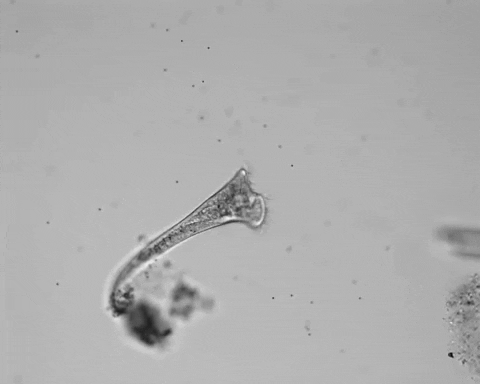
Jennings was studying S. roeselii, a member of a widespread genus of freshwater protist. These single cells are notable for their relatively large size and unique trumpet-shaped bodies. Their surfaces and trumpet “bells” are lined with hairlike projections called cilia, used to swim and to generate a vortex in the surrounding fluid, which sweeps food into their “mouths.” At the other end of their bodies, they secrete a holdfast, which attaches them to detritus to stay stationary while feeding.
With a microscope, a pipette and a steady hand, Jennings meticulously documented the behavior of S. roeselii when exposed to an environmental irritant in the form of carmine powder.
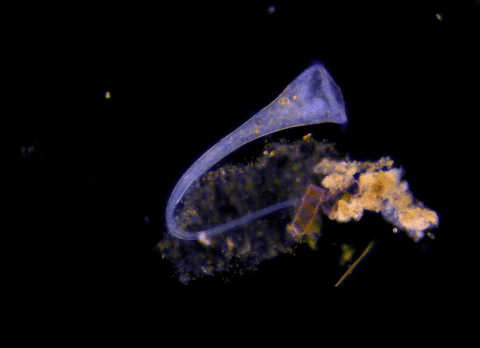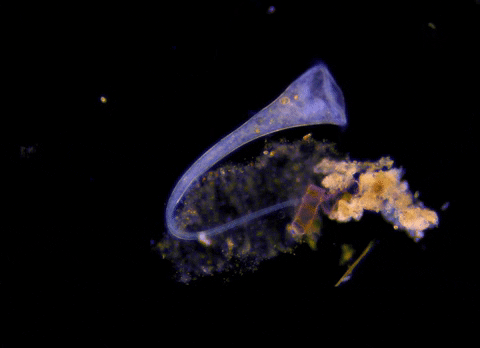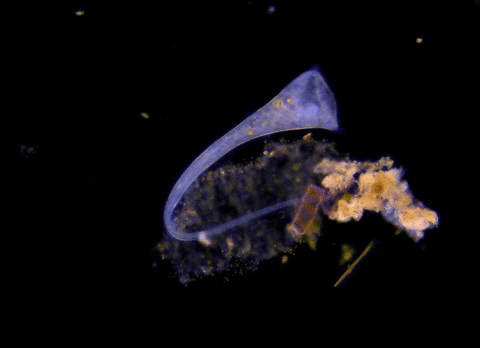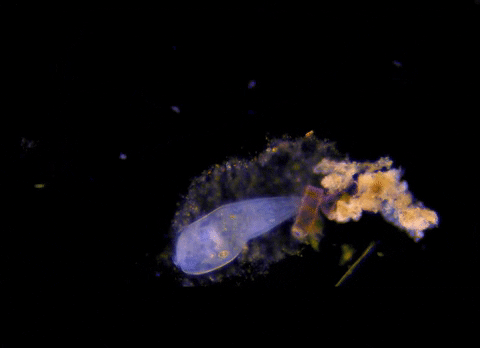
Jennings observed an ordered series of behaviors. He noted that, typically, S. roeselii would repeatedly bend its body to avoid the powder. If irritation persisted, it would reverse the movement of its cilia to expel particles away from its mouth. If this too failed, it would then contract, swiftly pulling itself down onto its holdfast like a barnacle retreating into its shell. Finally, if all prior efforts failed, S. roeselii would detach its holdfast and swim away.
These behaviors formed a hierarchy, an escalation of actions that the organism carried out based on a ranked preference. This observation suggested that it possessed some of the most complex behaviors known for a single cell with a single nucleus.
The experiment drew widespread interest, but subsequent efforts to replicate it—in particular, a study published in 1967—were unsuccessful. As a result, Jennings’ findings were largely discredited and forgotten by modern science.
Skunkworks project
Like carmine powder in an otherwise perfectly habitable puddle of water, this bothered Gunawardena, so he tracked down the 1967 study. To his astonishment, he found that the authors, who were unable to find S. roeselii, had used a different species to replicate Jennings’ experiment—Stentor coeruleus, which prefers to swim instead of attaching to feed.
Little surprise, then, that they failed to reproduce the results, Gunawardena thought. He became infatuated with trying to accurately replicate Jennings’ experiment. But as a mathematician by training running a medical school lab focused on molecular information processing, he found it difficult to convince others.
“I kept bringing up this idea at my lab group meeting, saying that it tells us something about the capabilities of single cells. We don’t think this way about how cells work anymore,” he said. “And, unsurprisingly, no one was interested. It’s ancient history, it’s descriptive biology—all the things young, bright trainees wouldn’t touch.”
But he persisted. One of his postdoctoral fellows, Sudhakaran Prabakaran, now a group leader at the University of Cambridge in England, became interested. And around eight years ago, Joseph Dexter, an undergraduate intern who later became Gunawardena's PhD student and who is now a fellow at the Neukom Institute for Computational Science at Dartmouth, was also attracted to the idea.
Driven only by an irrepressible sense of curiosity and history, with no formal grant support, the three engaged in a years-long side project.
“It was a completely off-the-books, skunkworks project,” Gunawardena said. “It wasn’t anyone’s day job.”
Dexter and Prabakaran designed and undertook the experiments, and their first challenge was finding S. roeselii. They hunted everywhere, even searching in local ponds. Ultimately, they located a supplier in England, which sourced the organisms from a golf course pond and shipped them across the Atlantic.
The team set up an experimental apparatus equipped with video microscopy and a micropositioning system to accurately deliver an irritant near the mouth of their S. roeselii test subjects. They initially used carmine powder but saw little response, and through trial and error, found that microscopic plastic beads were effective.
Lurking in the math
To their delight, the trio succeeded in eliciting—and reproducing—all the behaviors that Jennings once described.
However, they did not see the neat, orderly hierarchy of behaviors that Jennings had documented. Rather, there seemed to be considerable variation among subjects—one specimen might bend and alter its cilia before contracting, but another might only contract repeatedly, while another would alternate bending and contracting.
So, the three fell back on their core expertise as quantitative biologists. They developed a method to encode the different behaviors they saw into a series of symbols, and then used statistical analyses to look for patterns.
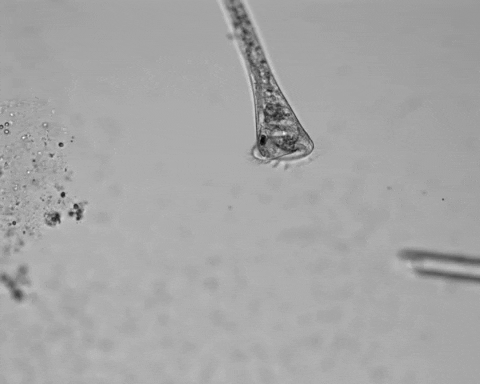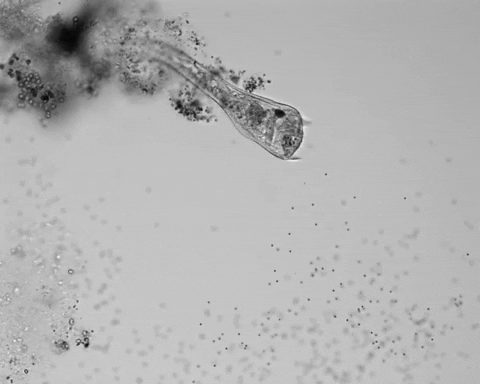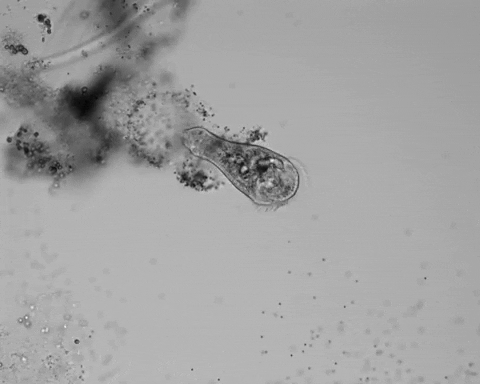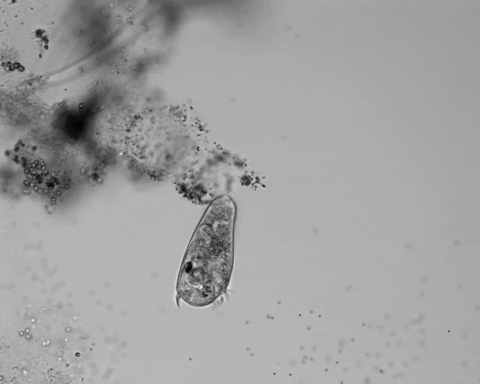
Where observation failed, math triumphed. There was, indeed, a behavioral hierarchy, the analysis revealed. When faced with an irritant, S. roeselii will, most of the time, begin by bending and altering its cilia, often simultaneously. If the irritation continues, it will then contract or detach and swim away. The latter behaviors almost always occur after the former, and organisms never detach without first contracting, indicating a preferred order of actions.
“They do the simple things first, but if you keep stimulating, they ‘decide’ to try something else. S. roeselii has no brain, but there seems to be some mechanism that, in effect, lets it ‘change its mind’ once it feels like the irritation has gone on too long,” Gunawardena said.
“This hierarchy gives a vivid sense of some form of relatively complex, decision-making calculation going on inside the organism, weighing whether it’s better to execute one behavior versus another,” he said.
Fair coin toss
In successfully replicating Jennings’ experiment and illuminating new quantitative observations about the behavioral capabilities of S. roeselii, the team hopes to have resolved the historical confusion about the accuracy of his findings.
But the results now raise numerous new questions.
The analyses showed that there is almost a perfectly even chance that any individual S. roeselii will choose to contract or detach, a clue that is particularly tantalizing to scientists who study how cells process information at the molecular level. The decision between the two behaviors is consistent, with each organism independently flipping an unbiased coin, regardless of previous actions, the authors said.
“It’s somehow basing its decisions, at the molecular level, on a fair coin toss,” Gunawardena said. “I can’t think of any known mechanism that would allow them to implement this. It’s incredibly fascinating and something Jennings never observed because we needed quantitative measurements to reveal it.”
More broadly, the authors say, the observation that single cells can be capable of complex behaviors could inform other areas of biology.
In developmental biology or cancer research, for example, the processes cells undergo are often referred to as programs, Gunawardena said, suggesting that cells are “programmed” to do what they do. “But cells exist in a very complex ecosystem, and they are, in a way, talking and negotiating with each other, responding to signals and making decisions.”
“I think this experiment forces us to think about the existence of, very speculatively, some form of cellular ‘cognition,’ in which single cells can be capable of complex information processing and decision-making in response,” he continued. “All life has the same underpinnings, and our results give us at least one piece of evidence for why we should be broadening our view to include this kind of thinking in modern biology research.”
“It also illustrates how, sometimes, we tend to ignore things not because they don’t exist, but because we don’t think it’s important to look at them,” he added. “I think that’s what makes this study so interesting.”
As part of his graduate studies, Joseph Dexter was supported by a National Science Graduate Research Fellowship (DGE1144152).
DOI: 10.1016/j.cub.2019.10.059
Image: Getty Images


