Since the early days of CRISPR-Cas9, researchers have known that this gene editing technology is excellent for breaking things. With precision, these scissor-like tools can be sent to any location in the genome to make a snip and break a gene. But exactly where and how CRISPR-Cas9 will disrupt the gene was anyone’s guess until now.
Harvard Medical School investigators at Brigham and Women’s Hospital, in collaboration with colleagues at the Broad Institute and MIT, have discovered that template-free Cas9 editing is predictable, and they have developed a machine-learning model that can predict insertions and deletions with high accuracy.
The team, led by co-corresponding authors Richard Sherwood, HMS assistant professor of medicine at Brigham and Women’s, David Liu, Harvard professor of chemistry and chemical biology and core institute member at the Broad, and David Gifford, visiting scholar in stem cell and regenerative biology at HMS and professor of computer science and engineering at MIT, has demonstrated that this approach can be used to edit and repair mutations related to three diseases in human cell lines—Hermansky-Pudlak syndrome, Menkes disease and familial hypercholesterolemia—with a predictable repair outcome in more than half of instances. These advancements have implications for both research and clinical applications. The team’s findings were published Nov. 7 in Nature.
“Like many projects, this one came out of a puzzling result: We wanted to use CRISPR-Cas9 to cause a random set of mutations at a particular spot in the genome, but we were finding that the mutations we were getting were far from random,” said Sherwood, a principal investigator in Brigham and Women’s Division of Genetics Research. “It turns out that the underlying sequence to which you are directing CRISPR-Cas9 allows you to predict, with a high degree of accuracy, which mutations you are most likely to get.”
Many genetic diseases arise from insertions and deletions that disrupt a gene’s function so being able to replicate these or fix them with accuracy would be a major coup. Conventional wisdom among CRISPR-Cas9 researchers has held that the tool randomly generates insertions and deletions in a gene unless researchers include a so-called repair template.
Sherwood’s team has found that even without a template, one can predict which insertions and deletions are most likely to occur. At certain genomic sites, one particular mutation dominates—the team used the term “precise-50” to indicate when a single such mutation comprised more than 50 percent of all major editing products.
To conduct their project, Sherwood and colleagues, including Christopher Cassa, HMS assistant professor of medicine and a principal investigator in the Division of Genetics Research at Brigham and Women’s, constructed a library of 2,000 Cas9 guide RNAs (gRNAs) paired with DNA target sites. They used this library to train inDelphi, a machine-learning model.
They found that inDelphi could predict deletions of varying lengths and single base pair insertions with high accuracy (r = 0.87) in five human and mouse cell lines, and that it predicted up to 11 percent of the gRNAs were precise-50.
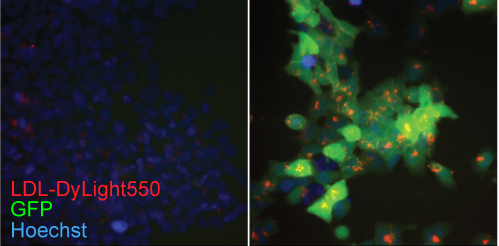
To confirm these findings, the team used select gRNAs to correct mutations in cells collected from patients with genetic diseases that result from microduplications—a chromosomal change in which a small amount of genetic material gets duplicated. Hermansky-Pudlak syndrome, especially common in Puerto Ricans, causes blood clotting deficiency and albinism. Menkes disease results in copper deficiency. The team also generated cells with microduplications found in patients to result in familial hypercholesterolemia, a disease in which LDL cholesterol levels are abnormally high. For all three diseases, delivering the appropriate Cas9 and guide RNA corrected the mutation with high efficiency.
The authors note that this work is still a proof-of-concept—while promising in cellular models in the lab, it requires further testing and additional steps to bring it into the clinic. In addition, only between 5 and 11 percent of Cas9 guide RNAs met the precise-50 standard.
Sherwood and the team will now work toward optimizing the efficiency of the guide RNAs by understanding why certain insertions or deletions are so much more common than others. They hope that others will leverage the tools they have developed to set regulatory standards for precision in existing therapeutic applications and expand what applications may be possible.
“Currently, most companies that are thinking about therapeutic applications for CRISPR-Cas9 are thinking about what genes you need to break to treat a disease. Our findings indicate that it’s possible to predict where we may be able to repair mutations instead,” said Sherwood. “We don’t want to settle for 50 percent mutation correction—we want to continue to improve. Now that we know that CRISPR-Cas9 editing is predictable, we have a parameter to measure how to fix disease mutations even more precisely.”
Funding for this work was provided by an NWO Rubicon Fellowship, National Science Foundation Graduate Research Fellowship, DARPA HR0011-17-2-0049, NIHRM1 HG009490L, R01 EB022376, R35 GM118062, Howard Hughes Medical Institute, 1RO1HG008363, 1R01HG008754, 1K01DK101684, the Human Frontier Science Program, NWO, National Science Foundation, Brigham Research Institute, Harvard Stem Cell Institute and American Cancer Society.
Adapted from a Brigham and Women’s news release.


