Image: Marcin Klapczynski, iStock / Getty Images Plus
Mother’s milk has been long touted for its salutary effects on the newborn and its ability to shield infants from certain infections.
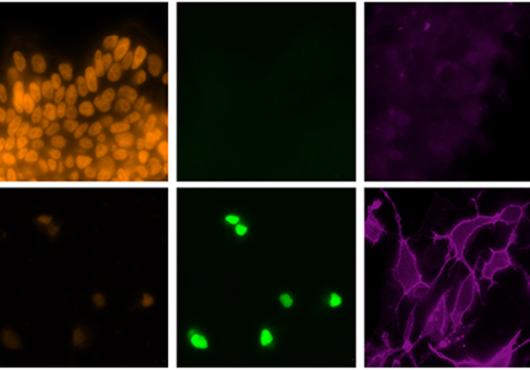
Now research from Harvard Medical School conducted in mice shows that at least part of its protective effects come from a surprising source: the microbes residing in maternal intestines.
Get more HM news
The new study, published Jan. 8 in Nature, shows that antibodies made in response to one particular organism in the maternal gut are passed on to the offspring both via milk and through the placenta to protect newborn pups from infection by at least one disease-causing, and potentially lethal, microbe, E. coli.
The findings add to a growing body of evidence pointing to the potent role the microbiota—the trillions of microbes that dwell in the gut, skin, mouth and other parts of the bodies of mammals, including humans—can play in disease and health.
But the new research goes a step further—it specifically identifies maternal microbiota as source of newborn immunity. It further suggests that the intestinal microbiota could offer immune protection even when mothers have had no prior encounters with an infection that allows them to build and pass on protective antibodies to their offspring.
“Our results help explain why newborns are protected from certain disease-causing microbes despite their underdeveloped immune systems and lack of prior encounters with these microbes,” said study senior investigator Dennis Kasper, professor of immunology in the Blavatnik Institute at Harvard Medical School. “Moreover, they raise the possibility that mothers can confer immune protection to their offspring even to pathogens that they haven’t themselves encountered in the past.”
If affirmed through further studies, the findings could inform the design of microbial therapies against dangerous infections such as E. coli and otherdisease-causing organisms, the researchers said.
“Albeit preliminary, we are hopeful these insights could inform the development of vaccines derived from commensal microbial molecules as a way to prevent infectious diseases,” said Kasper, who is also the William Ellery Channing Professor of Medicine at Brigham and Women’s Hospital. “Another therapeutic avenue could be the use of commensal microbes as probiotics that protect against diarrheal disease.”
Infectious diarrhea—most commonly due to E. coli or rotavirus—is the leading cause of malnutrition and the second leading cause of death globally in children under age 5. It causes 1.7 billion infections and claims more than 520,000 lives a year worldwide, according to the World Health Organization.
Without any prior exposure to microbes, a newborn’s immune system is a blank slate. For the first three weeks, a newborn’s immune protection is derived entirely from maternal antibodies passed onto the fetus during pregnancy via the placenta, during birth via the birth canal, and shortly after birth via breastfeeding.
In the current study, researchers worked with newborn mice genetically engineered to lack B cells—the antibody-producing factories of the immune system. Some of the newborn mice were subsequently raised by mothers that were also born without antibody-making B cells, and therefore, lacked protective antibodies. The other newborn mice were raised by mothers that had normal immune systems.
Mice exposed to protective antibodies from their mothers were far more resistant to E. coli infection than mice that were not exposed to such antibodies. It was as if the pups repelled the pathogen, the researchers observed. Indeed, their intestines had 33 times fewer E. colibacteria than newborn mice lacking maternal antibodies. By contrast, mice pups that were not exposed to protective antibodies developed disseminated E. colidisease.
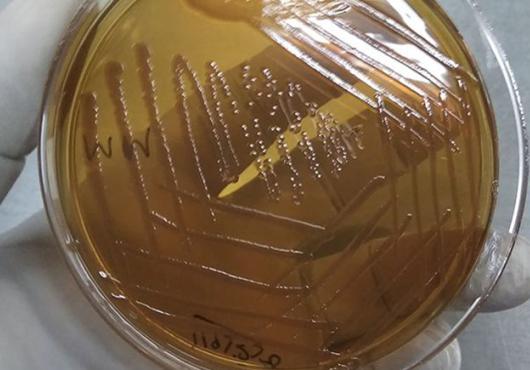
The researchers were also able to pinpoint the specific organism responsible for inducing the formation of the protective antibodies—a microbe called Pantoea,member of theEnterobacteriaceae bacterial family, which resides in the intestines of mice and other mammals, including humans.
Furthermore, the experiments showed that the antibodies enter both the intestines and bloodstream of newborns via the neonatal Fc receptor, a molecular channel on the placenta that helps ferry protective antibodies from the mother to the growing fetus. Thus far, the receptor has been known to transfer antibodies through the placenta. However, the experiments conducted in the new study show this receptor also absorbs antibodies derived from milk and ferries them from the intestines and into the bloodstream of the newborn mice, ensuring wider, systemic protection beyond the gut. Adult mice, in which this neonatal receptor loses its function with age, did not transfer protective antibodies from their gut to the bloodstream, the experiments showed.
Co-investigators included Wen Zheng, Wenjing Zhao, Meng Wu, Xinyang Song, Florence Caro, Ximei Sun, Francesca Gazzaniga, Giuseppe Stefanetti,Sungwhan Oh and John Mekalanos.
The workwas funded by the National Institutes of Health (NIAID Centers of Excellence for Translational Research grant no. 1U19 AI109764), with partial support from the European Union’s Research and Innovation Programme Horizon 2020 (Marie Skłodowska Curie grant agreement 661138).
Related:
Needle in a Haystack